How Microbiology contributes to the Pharmaceutical Industry?


微生物学到制药行业的最重要贡献是抗生素的发展。所有抗生素最初是微生物代谢的产物,然而最近的遗传操作使得生产更具增强的药物。
疫苗也是微生物学对药物发展的非常重要的贡献。对细菌疾病的疫苗产生通常需要大量细菌的生长。类固醇也可以从微生物中获得。
除了药物和生物产品的开发外,微生物学致力于制药实验室的质量控制。在药房指南下,预防药物,注射剂,滴眼液,鼻液和吸入产品的微生物污染。
Microbiological Test Methods
药物微生物测试分为几个类别。
The Growth Promotion test
生长促进试验是一个重要的品质control function in the pharmaceutical industry. It is imperative for establishing the ability and nutritive property of any media used to support growth when the inoculum contains a small number of microorganisms.
无菌测试。
Sterility testing is done on wide range of pharmaceutical products as parental preparations, ophthalmic & other non-injectable preparations, bulk solids and liquid solutions, antibiotic solids, and medical consumables and devices.
Microbial Limits Test
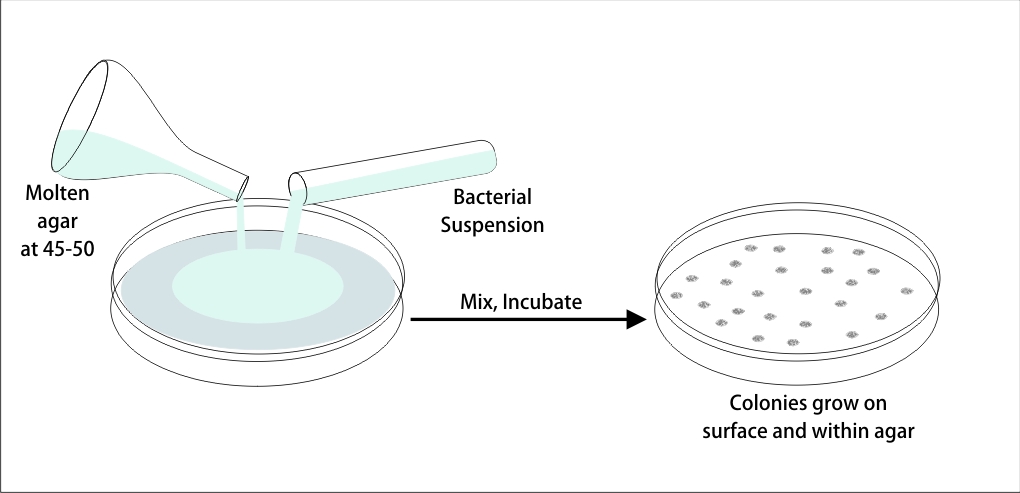
This test is used to estimate the total number of viable microorganisms or specific pathogens present in pharmaceutical products as tablets, capsules, oral suspensions, injectables, ophthalmic and nasal solutions and other medical devices.
基于该原理,当在提供生长培养基和有利的生长条件时,样品中存在的任何活的微生物细胞将产生单个菌落。
The enumeration of these colony-forming units (cfu) gives an estimate of the microbial population of the product. The microbial content of the product includes the total bacterial count (TBC), total yeast and mold count (TYMC). These tests are mandatory for the release of drug products.
Bioburden测试
原料和成品药品的生物矿石有助于确定产品是否符合美国药典的要求。Bioburden是在灭菌之前产品存在的微生物总数。
水测试
Water is one of the major commodities consumed by the pharmaceutical industry. Total viable count is studied to rule out microbial contamination. Tests for presence of coliforms,大肠杆菌and any other pathogens asPseudomonassp。Clostridia那沙门氏菌那Staphylococcus等等。
细菌内毒素(LAL测试)
内毒素是由革兰氏阴性细菌的细胞壁释放的天然化合物,其对人体可能有毒。这种材料是热原(导致人类的高烧伤)。细菌内毒素的测试用于使用素amoebocyte裂解物(LAL)来检测或定量内毒素,这是来自马蹄蟹的血细胞提取物(Limulus polyphemus的)
Drug quality and safety is the most important aspect of microbiological testing pharmaceutical products. The presence of any pathogenic bacteria, yeasts, moulds or bacterial toxins produced by microorganisms is strictly regulated to ensure prevention of any risk.
要点要记住
- 微生物用于生产抗生素,疫苗,类固醇等......
- 根据药长指南,严格遵循药品的微生物检测。
- Growth promotion tests establish the potential of any media to support growth when the inoculum contains a small number of microorganisms.
- 微生物极限测试和无菌测试用于鉴定产品的微生物负荷。





Fantastics! Please, I have desiring to go into vaccine production, I mean to study my masters in pharmaceutical microbiloogy. My area of specialization is clinical chemistry(medical laboratory science, Nigeria). I have 3.1 GP, the 2nd class lower division. And I want do my masters in pharm. Microbiology as I wan to be producing vaccines.
We absolutely love your blog and find the majority of your post’s to
be just what I’m looking for. Would you offer guest writers to write content available for you?
我不介意创建一个帖子或阐述
你在这里写的很多受试者。
Again, awesome website!
Yes, We would welcome contributions
Please be in touch with my colleague shanu[email protected]
Thanks & Regards !!
好的
简单的方式了解
Thanks
I am currently a 400 level microbiology student in the university of Lagos. I am in dire need of a seminar topic about pharmaceutical microbiology. All my topics of interest already exist. Please any suggestions for a new topic?
你好先生,它是来自尼泊尔的沙田帕尼。我也是生物技术的学生。希望您能帮助我向该领域提供知识。